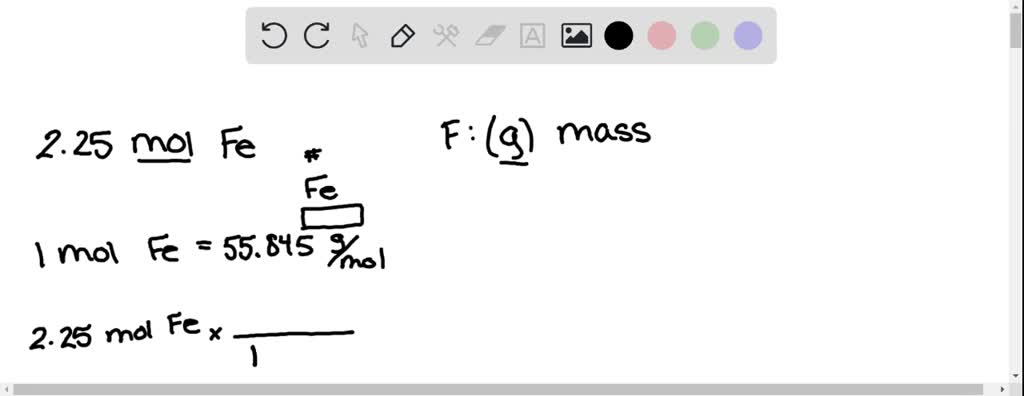
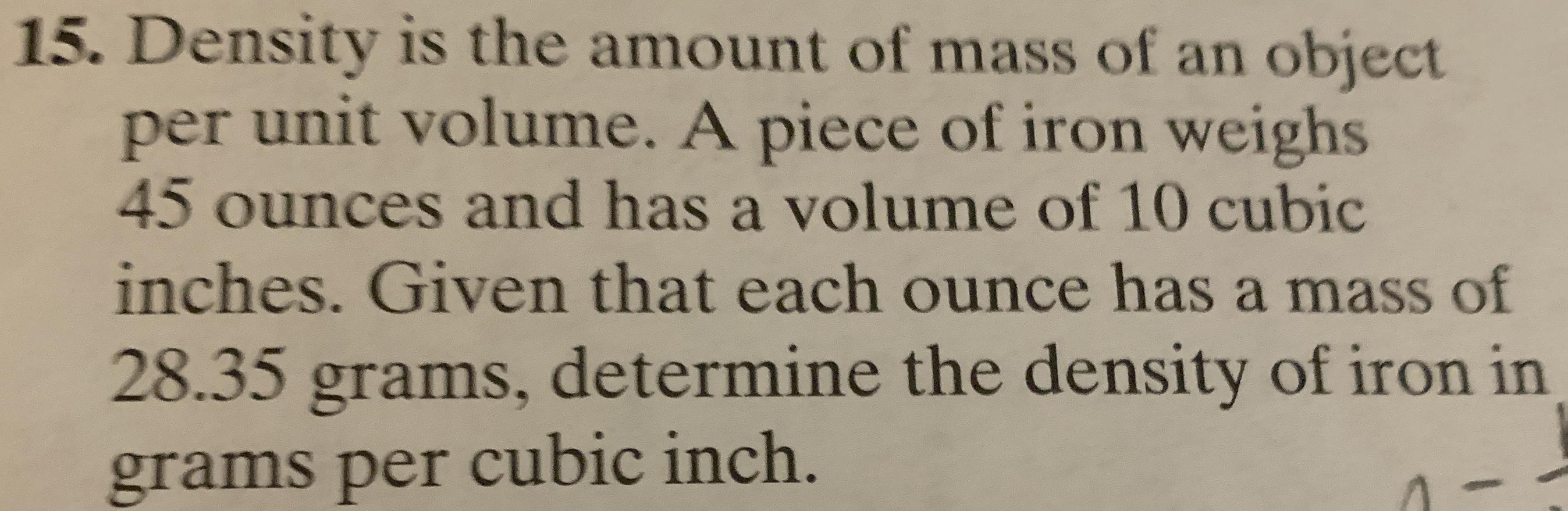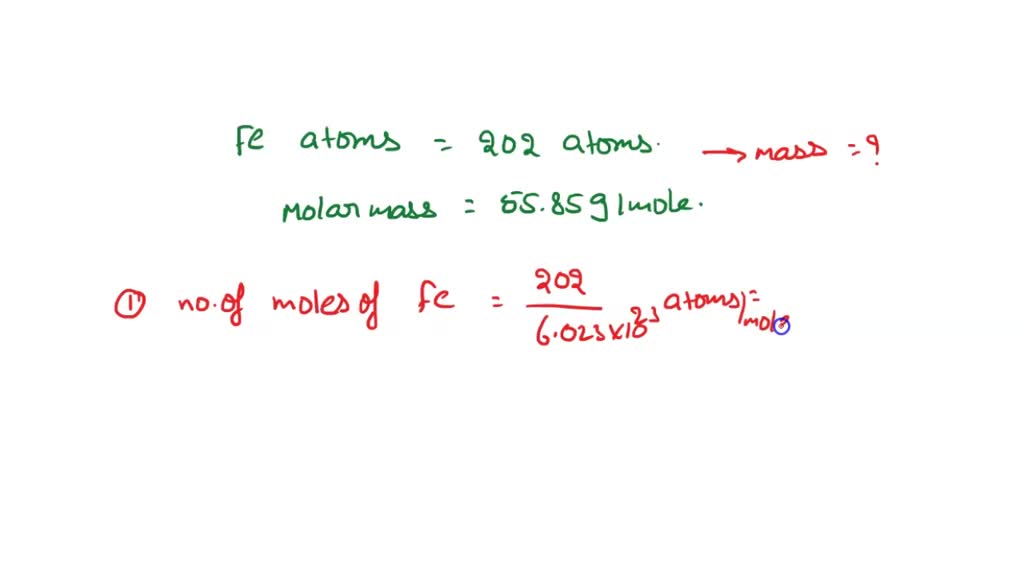Moles In Chemistry a mole is defined as 6.022x10 23 particles of a substance. Moles are not to be confused with this happy individual. This is a very special. - ppt download
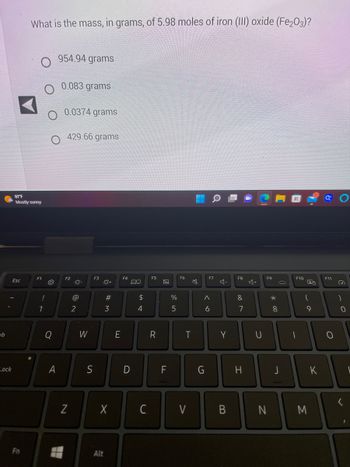
![Which has more number of atoms, 100 grams of sodium or 100 grams of iron. [Given, the atomic mass of NA = 23 u, Fe = 56 u ] Which has more number of atoms, 100 grams of sodium or 100 grams of iron. [Given, the atomic mass of NA = 23 u, Fe = 56 u ]](https://dwes9vv9u0550.cloudfront.net/images/2460098/1f33e3f8-015a-4d62-b26f-babbe7e573cd.jpg)
Which has more number of atoms, 100 grams of sodium or 100 grams of iron. [Given, the atomic mass of NA = 23 u, Fe = 56 u ]